Idiopathic Short Stature Market-By Treatment Type (Growth Hormone Replacement Therapy, Insulin like Growth Factor -1 (IGF-1) Therapy, Aromatase Inhibitors and low-dose Androgen Therapy) and By Geography - Forecast(2024 - 2030)
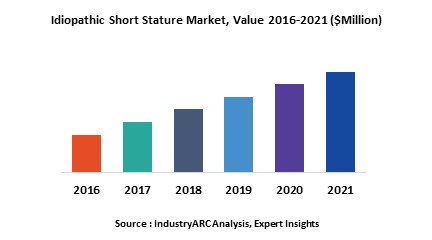
- Idiopathic Short Stature treatment market is segmented based on available treatment options. Although various treatment options are in the market majority of these are not approved for growth acceleration and hence they are considered under off label use. Recombinant growth hormone therapy, Insulin like Growth Factor -1 (IGF-1) therapy, Aromatase inhibitors and low-dose androgen therapy (with injectable testosterone and low dose androgen) are the available treatment options.
- This report is further segmented based on potential markets for Idiopathic Short Stature. A detailed analysis by region – North America, Europe, Asia Pacific (APAC) and Rest of the World (RoW), has been provided in this report.
- Eli Lilly
- Genentech/Roche
- Novo Nordisk
- Merck KGaA
- Pfizer
Key Market Players:
The Top 5 companies in the Idiopathic Short Stature Market are:
1 Roche
2. Merck Group
3. Eli Lilly and Company
4. Pfizer, Inc.
5. Novo Nordisk
List of Tables:
Table 1: Idiopathic Short Stature Market Overview 2023-2030
Table 2: Idiopathic Short Stature Market Leader Analysis 2023-2030 (US$)
Table 3: Idiopathic Short Stature Market Product Analysis 2023-2030 (US$)
Table 4: Idiopathic Short Stature Market End User Analysis 2023-2030 (US$)
Table 5: Idiopathic Short Stature Market Patent Analysis 2013-2023* (US$)
Table 6: Idiopathic Short Stature Market Financial Analysis 2023-2030 (US$)
Table 7: Idiopathic Short Stature Market Driver Analysis 2023-2030 (US$)
Table 8: Idiopathic Short Stature Market Challenges Analysis 2023-2030 (US$)
Table 9: Idiopathic Short Stature Market Constraint Analysis 2023-2030 (US$)
Table 10: Idiopathic Short Stature Market Supplier Bargaining Power Analysis 2023-2030 (US$)
Table 11: Idiopathic Short Stature Market Buyer Bargaining Power Analysis 2023-2030 (US$)
Table 12: Idiopathic Short Stature Market Threat of Substitutes Analysis 2023-2030 (US$)
Table 13: Idiopathic Short Stature Market Threat of New Entrants Analysis 2023-2030 (US$)
Table 14: Idiopathic Short Stature Market Degree of Competition Analysis 2023-2030 (US$)
Table 15: Idiopathic Short Stature Market Value Chain Analysis 2023-2030 (US$)
Table 16: Idiopathic Short Stature Market Pricing Analysis 2023-2030 (US$)
Table 17: Idiopathic Short Stature Market Opportunities Analysis 2023-2030 (US$)
Table 18: Idiopathic Short Stature Market Product Life Cycle Analysis 2023-2030 (US$)
Table 19: Idiopathic Short Stature Market Supplier Analysis 2023-2030 (US$)
Table 20: Idiopathic Short Stature Market Distributor Analysis 2023-2030 (US$)
Table 21: Idiopathic Short Stature Market Trend Analysis 2023-2030 (US$)
Table 22: Idiopathic Short Stature Market Size 2023 (US$)
Table 23: Idiopathic Short Stature Market Forecast Analysis 2023-2030 (US$)
Table 24: Idiopathic Short Stature Market Sales Forecast Analysis 2023-2030 (Units)
Table 25: Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 26: Idiopathic Short Stature Market By Treatment Type, Revenue & Volume, By Growth Hormone Replacement Therapy, 2023-2030 ($)
Table 27: Idiopathic Short Stature Market By Treatment Type, Revenue & Volume, By Insulin like Growth Factor -1 (IGF-1) Therapy, 2023-2030 ($)
Table 28: Idiopathic Short Stature Market By Treatment Type, Revenue & Volume, By Aromatase inhibitors and low-dose androgen therapy, 2023-2030 ($)
Table 29: North America Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 30: South america Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 31: Europe Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 32: APAC Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 33: Middle East & Africa Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 34: Russia Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 35: Israel Idiopathic Short Stature Market, Revenue & Volume, By Treatment Type, 2023-2030 ($)
Table 36: Top Companies 2023 (US$)Idiopathic Short Stature Market, Revenue & Volume
Table 37: Product Launch 2023-2030Idiopathic Short Stature Market, Revenue & Volume
Table 38: Mergers & Acquistions 2023-2030Idiopathic Short Stature Market, Revenue & Volume
List of Figures:
Figure 1: Overview of Idiopathic Short Stature Market 2023-2030
Figure 2: Market Share Analysis for Idiopathic Short Stature Market 2023 (US$)
Figure 3: Product Comparison in Idiopathic Short Stature Market 2023-2030 (US$)
Figure 4: End User Profile for Idiopathic Short Stature Market 2023-2030 (US$)
Figure 5: Patent Application and Grant in Idiopathic Short Stature Market 2013-2023* (US$)
Figure 6: Top 5 Companies Financial Analysis in Idiopathic Short Stature Market 2023-2030 (US$)
Figure 7: Market Entry Strategy in Idiopathic Short Stature Market 2023-2030
Figure 8: Ecosystem Analysis in Idiopathic Short Stature Market 2023
Figure 9: Average Selling Price in Idiopathic Short Stature Market 2023-2030
Figure 10: Top Opportunites in Idiopathic Short Stature Market 2023-2030
Figure 11: Market Life Cycle Analysis in Idiopathic Short Stature Market
Figure 12: GlobalBy Treatment TypeIdiopathic Short Stature Market Revenue, 2023-2030 ($)
Figure 13: Global Idiopathic Short Stature Market - By Geography
Figure 14: Global Idiopathic Short Stature Market Value & Volume, By Geography, 2023-2030 ($)
Figure 15: Global Idiopathic Short Stature Market CAGR, By Geography, 2023-2030 (%)
Figure 16: North America Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 17: US Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 18: US GDP and Population, 2023-2030 ($)
Figure 19: US GDP – Composition of 2023, By Sector of Origin
Figure 20: US Export and Import Value & Volume, 2023-2030 ($)
Figure 21: Canada Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 22: Canada GDP and Population, 2023-2030 ($)
Figure 23: Canada GDP – Composition of 2023, By Sector of Origin
Figure 24: Canada Export and Import Value & Volume, 2023-2030 ($)
Figure 25: Mexico Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 26: Mexico GDP and Population, 2023-2030 ($)
Figure 27: Mexico GDP – Composition of 2023, By Sector of Origin
Figure 28: Mexico Export and Import Value & Volume, 2023-2030 ($)
Figure 29: South America Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 30: Brazil Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 31: Brazil GDP and Population, 2023-2030 ($)
Figure 32: Brazil GDP – Composition of 2023, By Sector of Origin
Figure 33: Brazil Export and Import Value & Volume, 2023-2030 ($)
Figure 34: Venezuela Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 35: Venezuela GDP and Population, 2023-2030 ($)
Figure 36: Venezuela GDP – Composition of 2023, By Sector of Origin
Figure 37: Venezuela Export and Import Value & Volume, 2023-2030 ($)
Figure 38: Argentina Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 39: Argentina GDP and Population, 2023-2030 ($)
Figure 40: Argentina GDP – Composition of 2023, By Sector of Origin
Figure 41: Argentina Export and Import Value & Volume, 2023-2030 ($)
Figure 42: Ecuador Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 43: Ecuador GDP and Population, 2023-2030 ($)
Figure 44: Ecuador GDP – Composition of 2023, By Sector of Origin
Figure 45: Ecuador Export and Import Value & Volume, 2023-2030 ($)
Figure 46: Peru Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 47: Peru GDP and Population, 2023-2030 ($)
Figure 48: Peru GDP – Composition of 2023, By Sector of Origin
Figure 49: Peru Export and Import Value & Volume, 2023-2030 ($)
Figure 50: Colombia Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 51: Colombia GDP and Population, 2023-2030 ($)
Figure 52: Colombia GDP – Composition of 2023, By Sector of Origin
Figure 53: Colombia Export and Import Value & Volume, 2023-2030 ($)
Figure 54: Costa Rica Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 55: Costa Rica GDP and Population, 2023-2030 ($)
Figure 56: Costa Rica GDP – Composition of 2023, By Sector of Origin
Figure 57: Costa Rica Export and Import Value & Volume, 2023-2030 ($)
Figure 58: Europe Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 59: U.K Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 60: U.K GDP and Population, 2023-2030 ($)
Figure 61: U.K GDP – Composition of 2023, By Sector of Origin
Figure 62: U.K Export and Import Value & Volume, 2023-2030 ($)
Figure 63: Germany Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 64: Germany GDP and Population, 2023-2030 ($)
Figure 65: Germany GDP – Composition of 2023, By Sector of Origin
Figure 66: Germany Export and Import Value & Volume, 2023-2030 ($)
Figure 67: Italy Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 68: Italy GDP and Population, 2023-2030 ($)
Figure 69: Italy GDP – Composition of 2023, By Sector of Origin
Figure 70: Italy Export and Import Value & Volume, 2023-2030 ($)
Figure 71: France Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 72: France GDP and Population, 2023-2030 ($)
Figure 73: France GDP – Composition of 2023, By Sector of Origin
Figure 74: France Export and Import Value & Volume, 2023-2030 ($)
Figure 75: Netherlands Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 76: Netherlands GDP and Population, 2023-2030 ($)
Figure 77: Netherlands GDP – Composition of 2023, By Sector of Origin
Figure 78: Netherlands Export and Import Value & Volume, 2023-2030 ($)
Figure 79: Belgium Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 80: Belgium GDP and Population, 2023-2030 ($)
Figure 81: Belgium GDP – Composition of 2023, By Sector of Origin
Figure 82: Belgium Export and Import Value & Volume, 2023-2030 ($)
Figure 83: Spain Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 84: Spain GDP and Population, 2023-2030 ($)
Figure 85: Spain GDP – Composition of 2023, By Sector of Origin
Figure 86: Spain Export and Import Value & Volume, 2023-2030 ($)
Figure 87: Denmark Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 88: Denmark GDP and Population, 2023-2030 ($)
Figure 89: Denmark GDP – Composition of 2023, By Sector of Origin
Figure 90: Denmark Export and Import Value & Volume, 2023-2030 ($)
Figure 91: APAC Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 92: China Idiopathic Short Stature Market Value & Volume, 2023-2030
Figure 93: China GDP and Population, 2023-2030 ($)
Figure 94: China GDP – Composition of 2023, By Sector of Origin
Figure 95: China Export and Import Value & Volume, 2023-2030 ($)Idiopathic Short Stature Market China Export and Import Value & Volume, 2023-2030 ($)
Figure 96: Australia Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 97: Australia GDP and Population, 2023-2030 ($)
Figure 98: Australia GDP – Composition of 2023, By Sector of Origin
Figure 99: Australia Export and Import Value & Volume, 2023-2030 ($)
Figure 100: South Korea Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 101: South Korea GDP and Population, 2023-2030 ($)
Figure 102: South Korea GDP – Composition of 2023, By Sector of Origin
Figure 103: South Korea Export and Import Value & Volume, 2023-2030 ($)
Figure 104: India Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 105: India GDP and Population, 2023-2030 ($)
Figure 106: India GDP – Composition of 2023, By Sector of Origin
Figure 107: India Export and Import Value & Volume, 2023-2030 ($)
Figure 108: Taiwan Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 109: Taiwan GDP and Population, 2023-2030 ($)
Figure 110: Taiwan GDP – Composition of 2023, By Sector of Origin
Figure 111: Taiwan Export and Import Value & Volume, 2023-2030 ($)
Figure 112: Malaysia Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 113: Malaysia GDP and Population, 2023-2030 ($)
Figure 114: Malaysia GDP – Composition of 2023, By Sector of Origin
Figure 115: Malaysia Export and Import Value & Volume, 2023-2030 ($)
Figure 116: Hong Kong Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 117: Hong Kong GDP and Population, 2023-2030 ($)
Figure 118: Hong Kong GDP – Composition of 2023, By Sector of Origin
Figure 119: Hong Kong Export and Import Value & Volume, 2023-2030 ($)
Figure 120: Middle East & Africa Idiopathic Short Stature Market Middle East & Africa 3D Printing Market Value & Volume, 2023-2030 ($)
Figure 121: Russia Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 122: Russia GDP and Population, 2023-2030 ($)
Figure 123: Russia GDP – Composition of 2023, By Sector of Origin
Figure 124: Russia Export and Import Value & Volume, 2023-2030 ($)
Figure 125: Israel Idiopathic Short Stature Market Value & Volume, 2023-2030 ($)
Figure 126: Israel GDP and Population, 2023-2030 ($)
Figure 127: Israel GDP – Composition of 2023, By Sector of Origin
Figure 128: Israel Export and Import Value & Volume, 2023-2030 ($)
Figure 129: Entropy Share, By Strategies, 2023-2030* (%)Idiopathic Short Stature Market
Figure 130: Developments, 2023-2030*Idiopathic Short Stature Market
Figure 131: Company 1 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 132: Company 1 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 133: Company 1 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 134: Company 2 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 135: Company 2 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 136: Company 2 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 137: Company 3 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 138: Company 3 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 139: Company 3 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 140: Company 4 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 141: Company 4 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 142: Company 4 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 143: Company 5 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 144: Company 5 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 145: Company 5 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 146: Company 6 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 147: Company 6 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 148: Company 6 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 149: Company 7 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 150: Company 7 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 151: Company 7 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 152: Company 8 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 153: Company 8 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 154: Company 8 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 155: Company 9 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 156: Company 9 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 157: Company 9 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 158: Company 10 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 159: Company 10 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 160: Company 10 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 161: Company 11 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 162: Company 11 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 163: Company 11 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 164: Company 12 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 165: Company 12 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 166: Company 12 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 167: Company 13 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 168: Company 13 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 169: Company 13 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 170: Company 14 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 171: Company 14 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 172: Company 14 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
Figure 173: Company 15 Idiopathic Short Stature Market Net Revenue, By Years, 2023-2030* ($)
Figure 174: Company 15 Idiopathic Short Stature Market Net Revenue Share, By Business segments, 2023 (%)
Figure 175: Company 15 Idiopathic Short Stature Market Net Sales Share, By Geography, 2023 (%)
 Email
Email Print
Print

